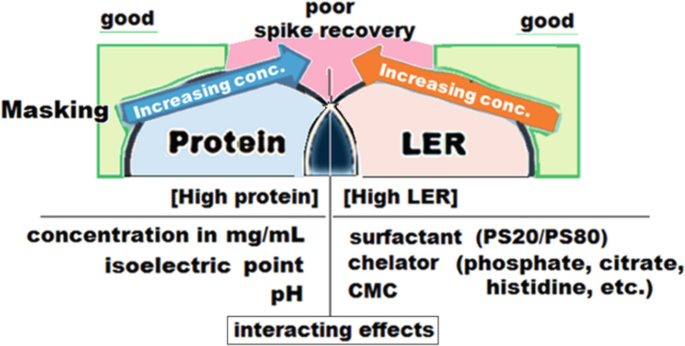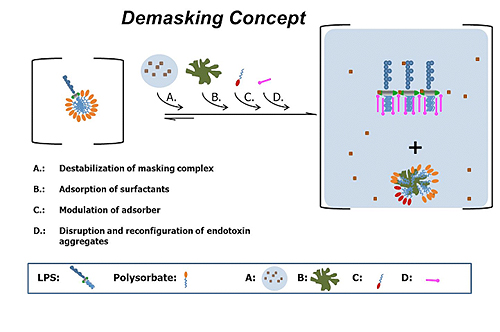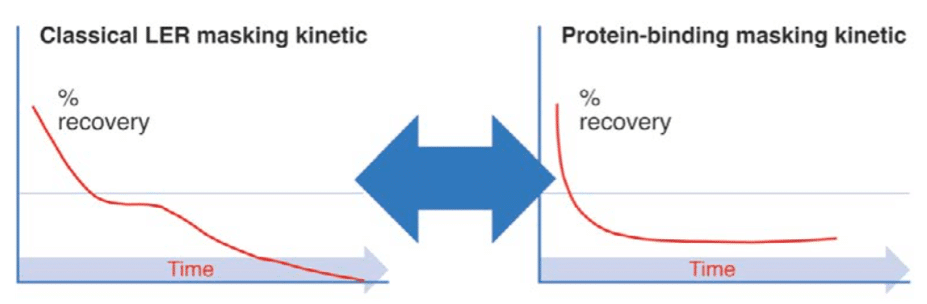
Endotoxin masking hold-time study parameter determination and performance - A3P - Pharmaceutical & Biotechnology Industry

Evidence Against a Bacterial Endotoxin Masking Effect in Biologic Drug Products by Limulus Amebocyte Lysate Detection | PDA Journal of Pharmaceutical Science and Technology
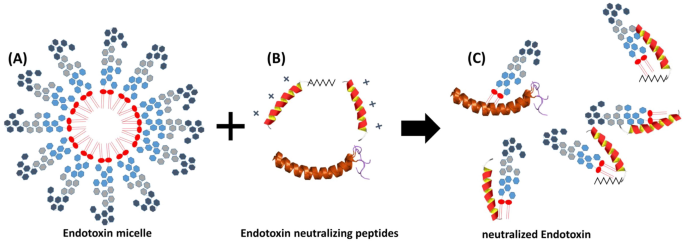
An in vitro study on factors affecting endotoxin neutralization in human plasma using the Limulus amebocyte lysate test | Scientific Reports

Fit For Purpose” Endotoxin Analytes | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

Removal of Endotoxin from rAAV Samples Using a Simple Detergent-Based Protocol: Molecular Therapy - Methods & Clinical Development

The Truth of Endotoxin Values - Points for Consideration During Investigation of Aberrant BET Results | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology
Bacterial Endotoxin Testing—Fast Endotoxin Masking Kinetics in the Presence of Lauryldimethylamine Oxide

Bacterial Endotoxin Testing: New Method for Unmasking Endotoxin in BiopharmaceuticalsBioProcess International


![PDF] Biotech Low Endotoxin Recovery : Beyond the Controversy | Semantic Scholar PDF] Biotech Low Endotoxin Recovery : Beyond the Controversy | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c1cb9dd0f9fba380d9009cbc645deeec89fbe127/2-Figure1-1.png)